Pharmaceutical marketing operates under a unique set of constraints that most other industries never have to consider. Every claim must be substantiated. Every piece of promotional material may need to pass through a medical-legal-regulatory (MLR) review. The audiences you are trying to reach, from healthcare professionals (HCPs) to payers to patients, each have distinct information needs, regulatory considerations, and preferred channels.
And yet, many pharma companies still approach marketing with outdated playbooks: heavy reliance on sales reps, one-size-fits-all messaging, and limited digital presence. The companies that are winning today are the ones building modern, integrated marketing strategies that combine scientific credibility with digital sophistication.
This guide provides a complete framework for building a pharmaceutical marketing strategy that works in 2026 and beyond. Whether you are a commercial team at an established pharma company or a marketing leader at a growing specialty pharma firm, this framework will help you prioritize your efforts and invest in the channels and tactics that drive measurable results. For context on how pharma marketing fits into the broader life sciences landscape, see our complete guide to life science marketing.
The Regulatory Reality of Pharma Marketing
Before diving into strategy, it is essential to understand the regulatory environment that shapes everything in pharma marketing. This is not a box to check at the end of a campaign. It is the foundation on which every marketing decision is built.
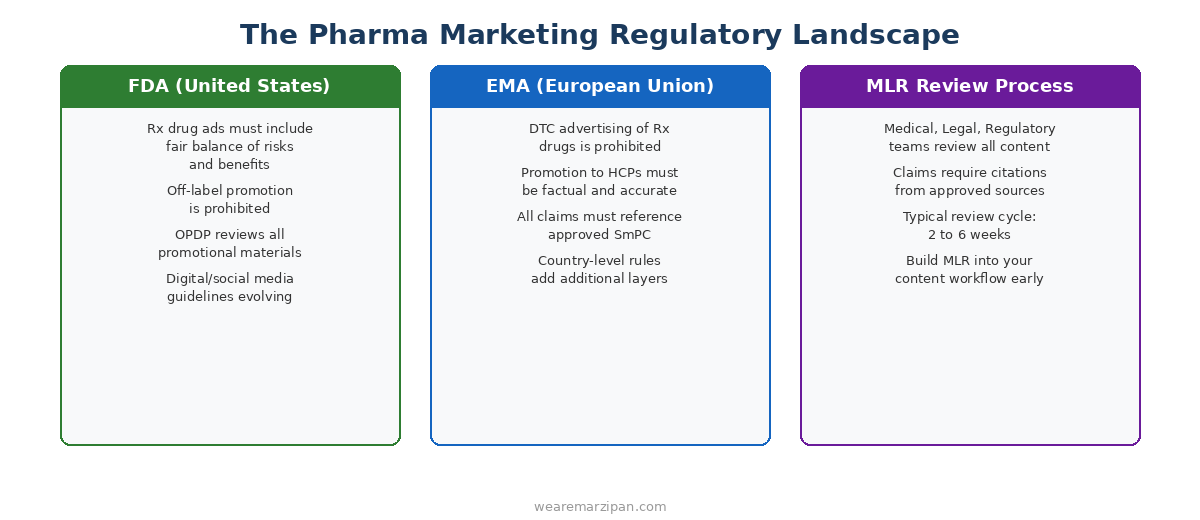
In the United States, the FDA's Office of Prescription Drug Promotion (OPDP) oversees pharmaceutical advertising and promotion. All promotional materials for prescription drugs must present a fair balance of both benefits and risks. Off-label promotion, meaning marketing a drug for uses not approved by the FDA, is prohibited. Digital and social media guidelines continue to evolve, and pharma marketers must stay current with the latest guidance documents.
In Europe, the regulatory landscape is even more restrictive in some ways. Direct-to-consumer (DTC) advertising of prescription drugs is prohibited across the EU. Promotion to healthcare professionals must be factual, accurate, and reference the approved Summary of Product Characteristics (SmPC). Individual EU member states often add their own additional regulations on top of EMA requirements.
For marketing teams, this means every piece of content, from a blog post to a conference banner to a LinkedIn ad, may need to go through an MLR review process. Medical, legal, and regulatory teams review all promotional materials to ensure compliance. This process typically takes two to six weeks, which has significant implications for your content production timelines. The smartest pharma marketing teams build MLR review into their content workflow from the start, rather than treating it as a last-minute hurdle.
The Pharmaceutical Marketing Strategy Framework
An effective pharma marketing strategy is not built around a single channel or tactic. It is an integrated system of six interconnected pillars, each reinforcing the others.
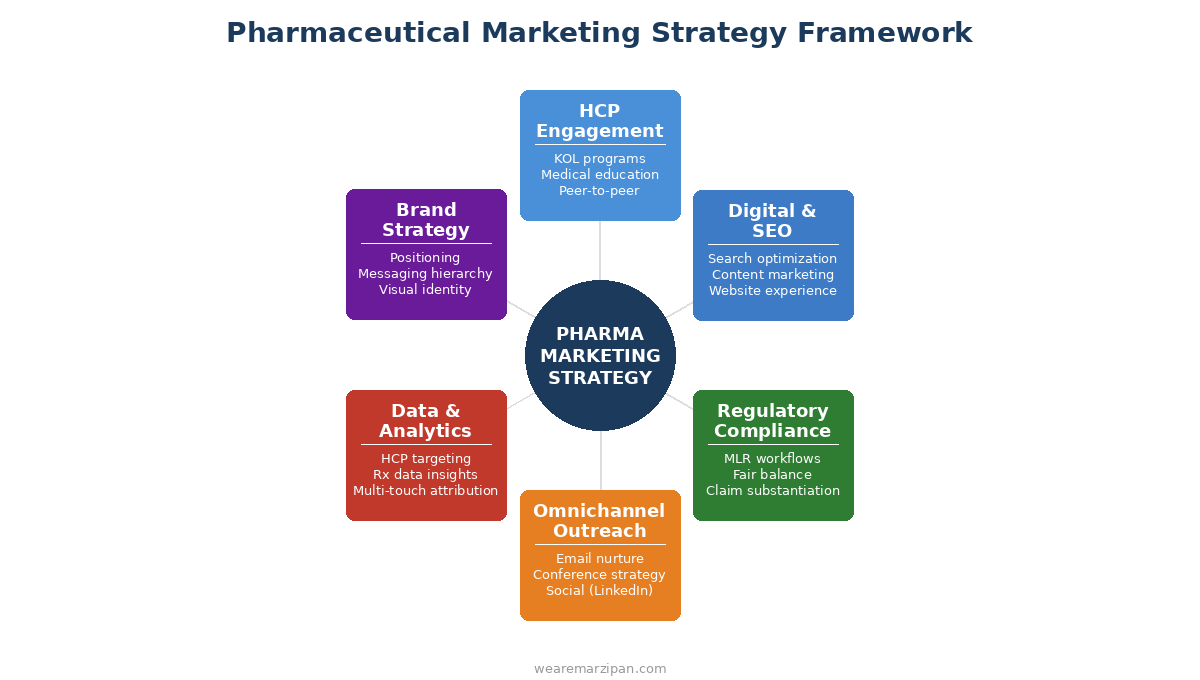
HCP Engagement
HCP marketing remains the cornerstone of pharmaceutical promotion. Healthcare professionals are the primary prescribers and influencers in the pharmaceutical value chain, and reaching them effectively requires a sophisticated, multi-layered approach.
Key opinion leader (KOL) programs are among the most powerful tools in pharma marketing. Identifying, engaging, and collaborating with respected physicians and researchers in your therapeutic area builds credibility that no amount of advertising can match. KOLs can serve as advisory board members, speakers at medical education events, authors of peer-reviewed publications, and advocates for your brand within their professional networks.
Medical education is another critical component. Continuing medical education (CME) programs, medical science liaison (MSL) activities, and peer-to-peer educational events help HCPs stay current on the latest clinical evidence and treatment options. When done well, these programs position your company as a trusted scientific partner rather than just another vendor.
The shift toward digital HCP engagement has accelerated dramatically. Physicians are increasingly researching treatments, reading clinical data, and engaging with pharma companies online. Your HCP engagement strategy needs to meet them where they are, combining in-person touchpoints with digital channels for a seamless experience.
Digital Marketing and SEO
Pharma digital marketing has gone from a nice-to-have to a strategic imperative. HCPs, payers, and patients all research health topics and treatment options online. If your company is not visible in those searches, you are ceding ground to competitors who are.
Search engine optimization is particularly valuable in pharma because the audiences are highly specific and the intent behind searches is clear. An oncologist searching for "PD-L1 inhibitor clinical data" or a hospital pharmacist searching for "biosimilar formulary considerations" knows exactly what they need. Ranking for these terms puts your content in front of the right person at the right moment.
Pharma content marketing requires a careful balance between being informative and staying compliant. The content that works best includes disease-state education that addresses unmet medical needs, mechanism of action explainers for HCPs, real-world evidence summaries and clinical data presentations, formulary and access resources for payers, and patient support and adherence resources. Each piece of content must be tagged with the appropriate regulatory classification (promotional vs. non-promotional) and routed through the correct approval pathway.
Your website is the hub of your digital presence. It should be organized to serve multiple audiences (HCPs, patients, payers) with clear navigation and gated access where required. For biotech companies earlier in their journey, many of these digital principles apply as well. Our biotech marketing guide covers the digital channel strategy in more detail.
How does your brand perform in AI search?
Run a free AEO audit to see how visible your brand is in ChatGPT, Perplexity, and Google AI Overviews.
Regulatory Compliance as a Strategic Advantage
Most pharma marketers view regulatory compliance as a constraint. The best ones view it as a competitive advantage. When you build compliance into your marketing workflow from the start, you move faster than competitors who treat it as an afterthought.
Practical steps to make compliance work for you, not against you:
- Create a content style guide that pre-addresses common MLR concerns: approved language for product claims, required fair balance statements, and citation formats. This reduces back-and-forth during review.
- Build an MLR-approved claims library. A centralized database of pre-approved claims and supporting references allows marketing teams to create new materials faster by assembling from approved building blocks.
- Invest in a promotional review platform like Veeva Vault PromoMats or similar tools that streamline the submission, review, and approval workflow.
- Involve MLR early. Bring your medical, legal, and regulatory reviewers into the content planning process, not just the review stage. When they understand the strategy behind a campaign, they can provide more constructive and efficient feedback.
Omnichannel Outreach
The days of relying solely on a sales force to drive pharmaceutical marketing are over. Modern pharmaceutical marketing requires an omnichannel approach that coordinates messaging across every touchpoint: in-person visits, email, conferences, digital advertising, social media, medical affairs activities, and patient support programs.
The key word is "coordinated." It is not enough to be present on multiple channels. The messaging, timing, and sequencing need to work together to create a cohesive experience. An HCP who sees your booth at a conference should receive a relevant follow-up email within 48 hours, not a generic newsletter three weeks later. A physician who downloads a clinical reprint from your website should be served related content on their next visit, not the same introductory material.
Email remains one of the most effective channels for HCP engagement. Segmented, personalized email campaigns that deliver clinically relevant content consistently outperform batch-and-blast approaches. LinkedIn is increasingly important for reaching HCPs in a professional context, particularly for disease-state education and thought leadership content. Conference marketing should be treated as a year-round strategy, not a one-week sprint, with pre-event awareness campaigns, on-site engagement, and systematic post-event follow-up.
Data and Analytics
Pharma has access to some of the richest data in any industry: prescription data (Rx), claims data, electronic health records, HCP engagement data, and digital interaction data. The challenge is connecting these data sources to create a unified view of your audience and measure marketing effectiveness.
The most advanced pharma marketing teams are building analytics capabilities around three core areas. First, HCP targeting and segmentation: using data to identify the highest-value prescribers and tailor outreach accordingly. Second, campaign measurement: tracking the impact of marketing activities on prescribing behavior, market share, and revenue. Third, multi-touch attribution: understanding which combination of touchpoints and channels drive the best outcomes across the long pharma sales cycle.
Privacy regulations add complexity. HIPAA in the US, GDPR in Europe, and various state-level privacy laws all affect how pharma companies can collect, store, and use data for marketing purposes. Compliance with these regulations is non-negotiable and should be built into your data strategy from the ground up, starting with proper consent management and cookie compliance.
Brand Strategy
Pharmaceutical branding operates at two levels: corporate brand and product brand. Your corporate brand represents your company's reputation, values, and therapeutic expertise. Your product brands each have their own positioning, messaging hierarchy, visual identity, and target audiences.
Strong pharmaceutical brands are built on three pillars: clinical evidence (does the product work?), patient outcomes (does it improve lives?), and trust (is the company behind it credible and transparent?). Every marketing activity should reinforce at least one of these dimensions.
Brand differentiation in pharma often comes down to the details. Two products in the same class may have similar efficacy data, but one company communicates its value proposition more clearly, supports HCPs with better educational resources, and provides a smoother patient experience. These are marketing problems, and they are where a strong pharma marketing strategy makes the difference between a market leader and an also-ran.
The Digital Transformation of Pharma Marketing
The pharmaceutical industry has historically been slower to adopt digital marketing than other sectors. That is changing rapidly. Several forces are driving this transformation.
HCP access has declined significantly over the past decade. Fewer physicians are willing to meet with sales reps in person, and many healthcare systems have restricted rep access entirely. Digital channels have become essential for reaching HCPs who are otherwise unreachable through traditional field force models.
Patient expectations have shifted. Patients are more informed, more engaged in their healthcare decisions, and more likely to research treatment options online before discussing them with their physicians. A strong digital presence helps pharma companies participate in these conversations and provide accurate, compliant information.
The pandemic accelerated digital adoption across the industry. Virtual conferences, remote detailing, digital HCP engagement, and telemedicine all became mainstream between 2020 and 2022, and most of these shifts have proven permanent. Pharma companies that invested in digital capabilities early are now reaping the benefits of that head start.
Measuring Pharmaceutical Marketing Success
Measuring the impact of pharmaceutical marketing requires a different approach than most B2B industries. The metrics that matter depend on your product lifecycle stage and your strategic objectives.
Pre-launch: Disease awareness, HCP education reach, KOL engagement, and market research insights. The focus is on building awareness and shaping the treatment landscape before product approval.
Launch: Market share uptake, new-to-brand prescriptions, HCP reach and frequency, and formulary wins. Drive adoption through multi-channel campaigns and access programs.
Growth: Market share, share of voice, patient adherence, and NPS among HCPs. Expand reach, deepen HCP relationships, and optimize your channel mix.
Mature / LOE: Brand loyalty, generic defense, patient retention, and cost efficiency. Maximize ROI from existing assets and prepare for lifecycle transitions.
The most common mistake in pharma marketing measurement is focusing exclusively on activity metrics (emails sent, events held, content published) rather than outcome metrics (prescriptions influenced, market share gained, pipeline built). Activity metrics tell you what you did. Outcome metrics tell you whether it worked.
Investing in proper marketing analytics infrastructure, including CRM integration, closed-loop reporting between marketing and sales, and multi-touch attribution modeling, pays dividends across every aspect of your marketing program. It helps you understand what is working, stop what is not, and continuously improve your return on marketing investment.
Building a Pharma Marketing Team (or Choosing an Agency)
Effective pharmaceutical marketing requires a blend of scientific knowledge, regulatory expertise, creative talent, and digital capabilities. Few companies have all of these skills in-house, which is why most pharma marketing organizations work with a combination of internal teams and specialized agency partners.
When evaluating pharmaceutical marketing companies or agencies, look for these qualities:
- Therapeutic area expertise. An agency that has worked in oncology may not be the right fit for a rare disease company. Look for relevant experience in your specific therapeutic area.
- Regulatory fluency. Your agency should understand the difference between branded and unbranded content, know how to navigate MLR review, and be familiar with the FDA and EMA guidance that applies to your promotional activities.
- Digital capabilities. Strong SEO, content marketing, email automation, and analytics skills are essential for modern pharma marketing. Ask about their approach to digital and how they measure performance.
- Scientific writing talent. The ability to translate complex clinical data into clear, compelling, and compliant marketing content is a rare skill. Review writing samples carefully.
- Integrated thinking. The best partners think across channels and audiences, not in siloed tactics. They should be able to connect HCP engagement with digital strategy with brand positioning into a cohesive program.
The Bottom Line
Building an effective pharmaceutical marketing strategy is one of the most complex challenges in B2B marketing. The regulatory constraints are real. The audiences are demanding. The sales cycles are long. And the stakes, both commercial and clinical, are extraordinarily high.
But the companies that get it right see outsized results. A well-executed pharma marketing program builds trust with HCPs, accelerates market access, improves patient outcomes, and drives sustainable revenue growth. It does this not through any single tactic, but through the disciplined integration of the six pillars outlined in this framework: HCP engagement, digital and SEO, regulatory compliance, omnichannel outreach, data and analytics, and brand strategy.
The pharmaceutical industry is in the middle of a significant digital transformation. The companies that invest in modern marketing capabilities now, rather than waiting until they are forced to change, will be the ones that lead their therapeutic areas for years to come. Start with the framework, build on the fundamentals, and iterate based on data. The opportunity is substantial for those willing to commit to the work.
Marzipan is a marketing agency built for life science and pharmaceutical companies. If you are looking for a partner who understands the regulatory landscape, speaks the language of science, and knows how to build marketing programs that drive commercial results, get in touch.

Written by
Joe Chamberlain
Head of Digital Marketing
Joe has over a decade of experience delivering high-impact digital strategies for B2B and B2C brands. He's built more than 200 websites and led countless SEO and performance marketing initiatives - each one focused on driving measurable ROI and sustainable growth.
More articles by Joe


